WHAT IS A HERNIA MESH?
A screen-like surgical implant used to repair hernias.
WHEN DOES A HERNIA OCCUR?
A hernia occurs when an organ, intestine or fatty tissue squeezes through a hole or a weak spot in the surrounding muscle or connective tissue. Hernias often occur at the abdominal wall. Sometimes a hernia can be visible as an external bulge particularly when straining or bearing down.
WHAT IS THE DIFFERENCE BETWEEN A LAPAROSCOPIC AND OPEN SURGERY?
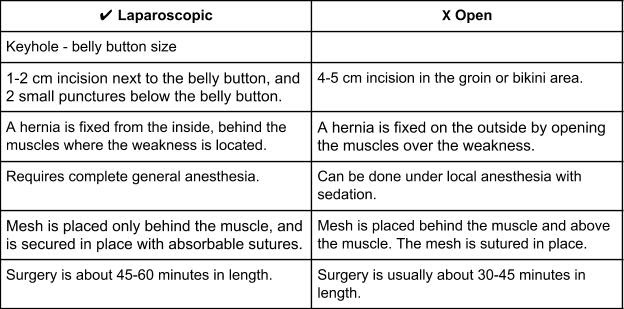
Laparoscopic Hernia Surgery Repair
A camera is placed through the muscle into the abdomen along with two small punctures for the operating instruments. From there, any scar tissue between the intestine and the hernia is cut allowing complete exposure of the hole.
A hernia mesh is rolled and placed through one of the laparoscopic ports into the abdomen. From there, it is pulled up against the muscle surrounding the hernia with stitches (sutures). Once the mesh is secured in place with about 4 sutures, a special stapling device is used to further fix the mesh to the healthy muscle.
WHAT DO HERNIA MESH LAWSUITS ALLEGE?
- Designing an “unreasonably dangerous and defective product” with an unreasonable risk of severe adverse reactions
- Selling a product that failed to reasonably perform as intended and was directly responsible for the necessity of surgical removal
- Failure to warn defendants and medical professionals of the risks associated with the mesh products.
WHICH HERNIA MESHES HAVE BEEN RECALLED BY THE FDA?
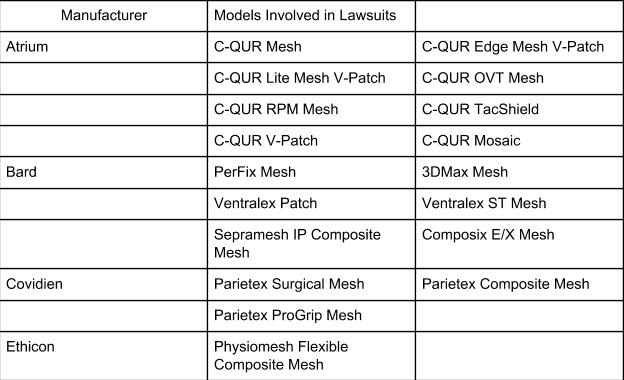
Atrium: Recalled by the FDA in October 2012 for their failure to address C-Qur mesh infection and sterility complaints. 35 complaints were made after finding human hair in the device. The FDA filed a lawsuit against Atrium for poor quality control at their New Hampshire manufacturing facility, forcing the company to stop production and distribution of the C-Qur mesh.
Ethicon’s Physiomesh (Johnson & Johnson Subsidiary)Recalled by the FDA in July 2016. The company voluntarily issued the recall due to a higher risk of recurrence of revision surgery compared to other brands.
WHAT IS THE STATUS OF CURRENT HERNIA MESH LITIGATIONS?
As of January 2019, nearly 3000 lawsuits were pending in federal MDLs against three hernia mesh manufacturers.
Atrium C-QUR Mesh MDL
A total of 1,025 Atrium C-QUR mesh lawsuits were pending in New Hampshire District Court as of January 2019. The Judicial Panel on Multidistrict Litigation established the MDL in December 2016.
Ethicon Physiomesh Flexible Composite Mesh MDL
As of January 2019, there were 1,696 lawsuits over Ethicon’s Physiomesh Flexible Composite Hernia Mesh pending in Georgia Northern District Court. The MDL panel established the MDL in June 2017.
The first trial dates are set for September 16, 2019.
Bard Polypropylene Hernia Mesh MDL
As of January 2019, there were 233 lawsuits over Bard Polypropylene Hernia Mesh pending in the Southern District of Ohio.
HERNIA MESH MAKERS AND PRODUCTS IN CURRENT LAWSUITS
WHO IS ELIGIBLE FOR A HERNIA MESH LAWSUIT?
and had or have scheduled a hernia revision surgery